Water Quality Data for the Lower Merrimack River
Continuous monitoring station in the Lawrence section of the river
 Water monitor in the Lawrence section of the Lower Merrimack River
Water monitor in the Lawrence section of the Lower Merrimack River
Water monitor in the Lawrence section of the Lower Merrimack River
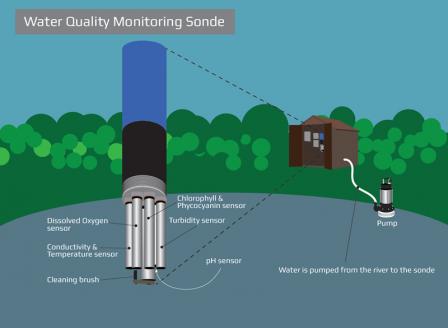
Every 15 minutes, from December 2016 through November 2018, this monitoring station took measurements for temperature, dissolved oxygen, pH, specific conductance, turbidity, chlorophyll, and phycocyanin. The data was transmitted in real-time using telemetry for remote access. The data from this station is used to assess water quality conditions.
Data from the monitoring station are presented below.
Learn more about the structure of the monitoring station.
Data Stream
The data presented on this website is considered preliminary data and may be subject to future revision or qualifiers. The data for this site was transmitted directly from the instrument with no or little review. Inaccuracies may be presented due to instrument malfunction or physical changes at the monitoring site. Prior to water quality measurements, the water flows through a pump and a course filter which may slightly alter the measurements.
Click on each of the eight types of measurements above to display that measurement. Click again to hide.
Data View #2 - View the live data in another graph that allows you to compare parameters to their associated water quality standards.
Temperature
Water temperature is measured in degrees Celsius (°C) which can be converted to degrees Fahrenheit (°F) by multiplying by 9, dividing by 5, and then adding 32. The Commonwealth of Massachusetts has established protective water quality standards for warm water fisheries in Class B waters. Water is not to exceed 28.3°C (83°F).
Temperature controls many biological and chemical processes that take place in the water. If water gets too warm, it can harm fish and aquatic life, influence undesirable changes in biological communities and increase the likelihood of algal blooms.
Humans can increase the water temperature by:
- Removing trees and therefore shade from the river
- Building structures that prevent infiltration and increase the temperature of stormwater run parking lots and black roofs
- Discharging heated water from Power Plants into the River. Power plants' discharges are regulated to have minimum impact on fish and other organisms.
Specific Conductance (Conductivity)
Specific conductance is a measure of the ability of water to pass an electrical current. Specific conductance measurements are affected by the presence of dissolved solids such as salts. For example, sea water will have higher specific conductance and distilled water will have lower specific conductance. Specific Conductance is reported at 25 degrees Celsius (°C), and reported in millisiemens per centimeter (mS/cm).
Road salt used during winter storms can drain into waterways. The Lower Merrimack's proximity to major highways from Manchester to Lawrence, along with harsh New England winters, makes increased contamination from road salt likely. Road salt is measured by specific conductance. Excessive road salt applications may lead to measurements that exceed water quality standards for fresh water aquatic life, such as freshwater fishes.
pH
To determine if water is acidic or basic pH is used. The range of pH goes from 0 - 14 Standard Units (S.U.), with 7 being neutral. A pH of less than 7 indicates acidity, whereas a pH of greater than 7 indicates a basic or alkaline condition. pH is reported in logarithmic units and each whole number represents a 10-fold change in the acidity/basicity of the water. Water with a pH of 5 is ten times more acidic than water having a pH of 6. Since pH can be affected by chemicals in the water, pH is an important indicator of water that is changing chemically.
The Commonwealth of Massachusetts has established protective water quality standards for Class B waters. Class B waters are those that the state has determine should be suitable for fishing and swimming. Those standards indicate pH should be in the range of 6.5 through 8.3 and not more than 0.5 units outside of the natural background range.
Pollution can change the water's pH, which in turn can harm organisms living in the water. Lower pH readings are sometimes seen in fresher waters due to acid precipitation or even naturally-occurring organic acids, which can be found in bogs and some wetlands. High pH readings can occur during algae blooms due to chemical processes associated with photosynthesis.
Dissolved Oxygen
Dissolved Oxygen (DO) measures how much oxygen is dissolved in the water. This is very important to fish and aquatic life that depend on an adequate supply of DO to live. DO is measured in milligrams per liter (mg/L) or percent saturation (%). Different organisms require different levels of DO. Generally, DO is lower at deeper depth where less mixing occurs with the surface water and where water becomes stratified.
The Commonwealth of Massachusetts has established protective water quality standards for warm water fisheries in Class B waters. The standards indicate that DO levels should not be less than 5 mg/L. When concentrations go below this level, sensitive organisms, such as some fish, may become stressed or die, especially if exposed to these conditions for prolonged periods.
Several factors can affect the concentration of dissolved oxygen:
- Temperature reduces the levels of dissolved oxygen since warmer water cannot dissolve as much oxygen as colder water.
- Algae and other aquatic plants also have an effect on dissolved oxygen. How?
- During the daylight, when the algae are photosynthesizing, they can produce more oxygen than is able to escape into the atmosphere, thus leading to short-term levels that can be greater than 100% (super saturated). At night time algae respire and consume oxygen. These daily swings can be quite large when there are algae blooms fueled by nutrient pollution, and can lead to poor aquatic conditions and fish kills.
- When the algae dies or when other organic matter enters the system and decomposes, dissolved oxygen is consumed.
Turbidity
Turbidity is one of the ways to determine water clarity by measuring of how much suspended material is in the water. Suspended materials can include soil particles (clay, silt, and sand), algae, plankton, microbes, and other substances. Turbidity is measured in Formazin Nephelometric Units (FNUs). Under normal condition, turbidity values are usually less than 10 FNUs at the Lower Merrimack.
Higher turbidity reduces the amount of light penetrating the water. Suspended materials can clog fish gills, reducing resistance to disease in fish, lowering growth rates, and affecting egg and larval development. As the particles settle, they can blanket the bottom, especially in slower moving waters, and smother fish eggs and benthic macroinvertebrates. Sources of turbidity include: urban runoff, soil erosion, waste discharge, eroding stream banks, excessive algal growth, dredging or anything that would re-suspend bottom sediments.
Chlorophyll
Chlorophyll is a green pigment found in plants, algae and cyanobacteria. Chlorophyll is extremely important in the photosynthesis process, which allows plants to absorb energy from light. Chlorophyll is measured using a fluorescence sensor.
Some levels of chlorophyll and algae are healthy and provide the base of the food web. When too many nutrients enter the water, algae and cyanobacteria concentrations can take off and form "blooms." Blooms can be unhealthy for other organisms in the water, people, and pets in contact with the water. Chlorophyll is measured in Raw Fluorescent Units (RFUs).
Phycocyanin
Phycocyanin is a pigment found in cyanobacteria that is measured in freshwater environments. Phycocyanin is measured using a fluorescence sensor. Phycocyanin is measured in Raw Fluorescent Units (RFUs).
The phycocyanin measurement is used as a field screening for cyanobacteria and cyanobacteria blooms, also called blue green algae. When blooms are formed, the risk of toxin contamination of surface waters increases. Cyanobacteria, also known as blue-green algae, are of special concern because of their potential impacts on drinking and recreational waters. When a bloom is occurring, people should avoid contact with the water and keep their pets out of the water.
